Low DNA Taq HS 5 U/µL
Low DNA Taq HS, 5 U/µL is a highly purified chemically modified hot start Taq DNA polymerase and buffer system, with separate magnesium. It has been developed for low bioburden and very high sensitivity over a wide range of PCR templates.
Have questions about a product?
Contact us to learn more about Meridian’s molecular or immunoassay reagent portfolio. We want to hear from you!
High sensitivity and specificity using Low DNA Taq HS
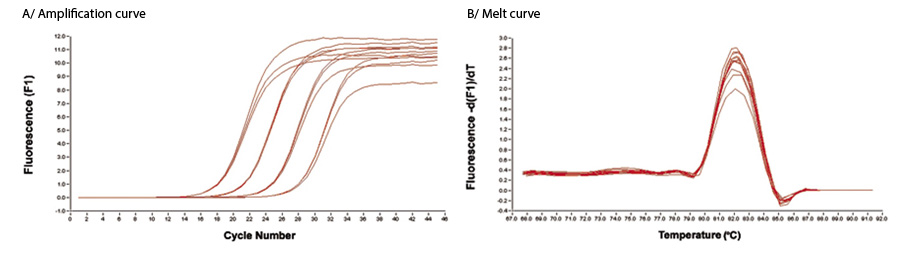
A 10-fold dilution of DNA was used in a qPCR assay using Low DNA Taq HS and an intercalating dye. Each dilution was run in triplicate using standard reaction conditions. A/ The amplification curve demonstrates the reproducibility and sensitivity of the Low DNA Taq HS and B/ The single distinct peak in the melt curve illustrates the products are a single discrete species, with no additional bands or primer/dimers.
Low DNA Taq HS, 5 U/µL, MDX009
Available in 0.1 mL (500 Units) or 10 mL (50,000 Units) aliquots
Description
A chemical moiety is attached to Low DNA Taq HS, 5 U/µL making it inactive at ambient temperature, avoiding extension of non-specifically annealed primers or primer dimers and providing higher specificity of DNA amplification. Low DNA Taq HS, 5 U/µL catalyzes 5′-3′ synthesis and possesses low 5′-3′ exonuclease activity, however these are not detectable before activation, enhancing PCR sensitivity. The functional activity of the enzyme is restored during a 10-minute incubation at 95 °C. The low DNA background and stringent hot-start properties of Low DNA Taq HS, 5 U/µL are ideal for PCR of low-copy bacterial targets and avoiding false-positive amplification, such as in water testing.
Specifications
| Description | A highly purified Taq DNA polymerase. The low DNA background and stringent hot-start properties are ideal for PCR of low-copy bacterial targets and avoiding false-positive amplification, such as in water testing. |
| Concentration | 5 U/µL |
| Appearance | Clear, colorless solution |
| Hot Start | Chemical |
| Purity | >90% |
| Application | PCR, two-step RT-PCR |
| Presentation | 1 vial |
| Storage | -20 °C |
| Mix stability | See outer label |
| Consistency | ±0.5 Ct variance between test and reference sample |
| DNA Contamination | None detected in PCR amplification with traces overlay with the negative control on E. coli and mouse genomic DNA specific targets. |
| DNase Contamination | No detectable degradation |
Catalogs & Brochures
Reagent Solutions for Molecular DiagnosticsReagent Solutions for Molecular Diagnostics
FAQs: Low DNA qPCR mix
With a chemical hot start Taq polymerase, the polymerase is covalently linked with chemical groups to block enzyme activity at room temperature.
Advantages
• More stringent than antibody hot-start methods, so there is no activity even for long periods at room temperature, minimizes non-specific amplification
• Low bioburden, as it is free of animal and bacterial-origin components
Considerations
• Longer activation time required for the polymerase to become fully active
Low DNA Taq HS, 5 U/µL lacks a 3′ – 5′ exonuclease activity. However, the enzyme does have 5 ‘ – 3′ nuclease activity. During the extension step of a PCR amplification, the enzyme will hydrolyze any blocking strand starting from its 5’ end.
Low DNA Taq HS, 5 U/µL lacks proofreading activity, so it will leave a 3′-overhang which can be used with TA cloning. In order to drive the reaction to the extra A state, a final extension time at 72°C should be increased to 15-30 minutes.
Yes, once activated, Low DNA Taq HS, 5 U/µL remains active. Lowering the temperature will not inactivate Low DNA Taq HS, 5 U/µL.
No the 10-minute activation will the enzyme, it will remain fully functional during the subsequent cycling.
Multiplex PCR involves the co-amplification of multiple amplicons in a single PCR. Since several sets of primers are being added to a single reaction, the potential for primer dimer formation or mispriming and so loss of specificity and so a decrease in yield of specific product exists. Because Low DNA Taq HS, 5 U/µL remains active has no activity prior to the hot start PCR, the multiple primers do not have the possibility to react with themselves, dramatically increasing specific product yield.
Get In Touch With A Specialist
Have questions about a product? Want to learn more about Meridian’s molecular or immunoassay reagent portfolio? We want to hear from you!