Lyo-Ready™ Genotyping Direct qPCR Stool
Lyo-Ready™ Genotyping Direct qPCR Stool has been optimized to provide highly specific allelic discrimination as demonstrated by excellent cluster separation, even in the presence of PCR inhibitors found in crude human stool samples. Furthermore, it can be used in a liquid or lyophilized format to create ambient-temperature stable assays, making it ideal for point-of-care (POC) devices.
Have questions about a product?
Contact us to learn more about Meridian’s molecular or immunoassay reagent portfolio. We want to hear from you!
20 mg/mL Human Stool and Gastric Cancer-Associated p53 Target
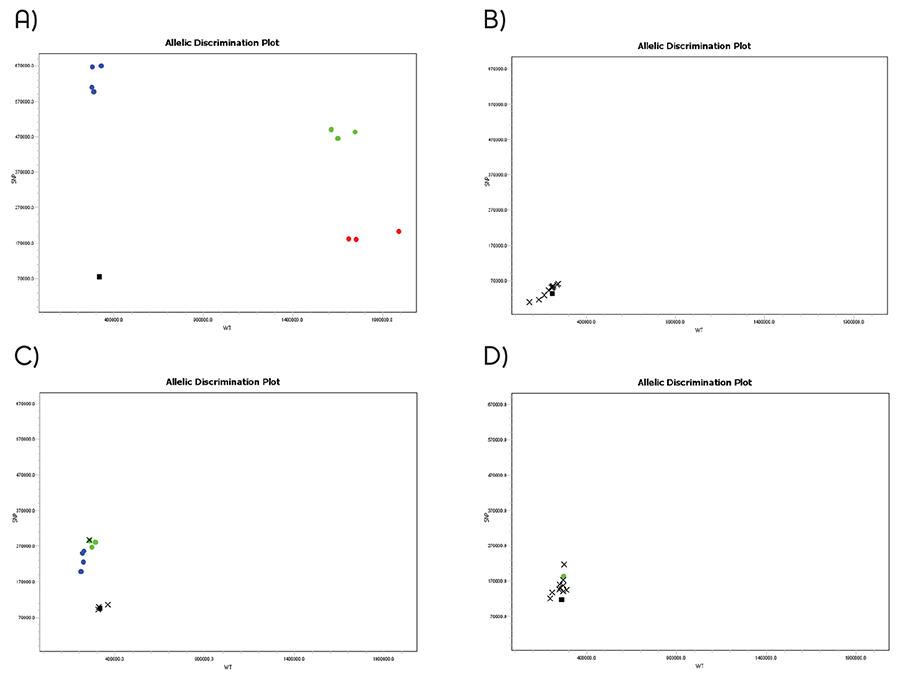
Detection of colorectal cancer-associated p53 mutation (SNP accession number: rs1042522) was performed using A) Lyo-Ready™ Genotyping Direct qPCR Stool, B) Kapa Probe Force, C) TaqPath™ and D) Type-it Fast Kits, in presence of 20 mg/mL human stool. Homozygous Allele C (red) and allele G (blue) and heterozygous samples for allele C/G (green) were compared with a NTC (black) and x for undetermined. The results illustrate the ability of Lyo-Ready™ Genotyping Direct qPCR Stool to form tight clusters and to achieve accurate allelic discrimination of clinically relevant mutations directly from crude human stool samples.
Lyo-Ready™ Genotyping Direct qPCR Stool, MDX148
Available in 1,000 Unit or 10,000 Unit aliquots
SNPs (single nucleotide polymorphisms) or point mutations are the most common types of genetic variation and comprise the major part of phenotype diversity between individuals. SNPs can be responsible for both resistance or susceptibility to a certain disease and once individual SNPs have been identified by sequencing, they can be used for pharmacogenetics, in evaluating and predicting a patient’s response to treatment and risk of adverse events, or for diagnostics, such as in cancer screening. Clinical screening of SNPs requires large-scale multiplexed genotyping, which traditionally relies on DNA extraction from complex samples such as blood, to achieve sensitive DNA amplification. However, blood collection is invasive and requires a professional, stool on the other hand is non-invasive, safe, less expensive and easy to do at home, making it ideal for POC and frequent sampling.
Meridian’s new Lyo-Ready Genotyping Direct qPCR Stool is a mix that is designed for fast, precise, and reproducible allelic discrimination and cluster separation. Its advanced formulation enables highly sensitive amplification of SNPs and other point mutations from stool samples and overcomes inhibitors such as bile salts that can damage DNA and inhibit PCR reactions. Furthermore, it can be used in a wet format or lyophilized to create ambient-temperature stable assays, making it ideal for point-of-care (POC) devices.
Catalogs & Brochures
Lyo-Ready Genotyping Direct qPCR StoolLyo-Ready Genotyping Direct qPCR Stool
Lyo-Ready Direct DNA and RNA/DNA LAMP Stool Master MixesLyo-Ready Direct DNA and RNA/DNA LAMP Stool Master Mixes
Meridian Air Dryable Direct StoolAir-Dryable™ Direct StoolCatalog
FAQs: Lyo-Ready Genotyping Direct qPCR Stool
Allelic discrimination are methods to discriminate between two or more sequence changes at a particular gene locus. Typically, one allele (“wild type” DNA sequence) is common, and other alleles (mutation) is rare. In a SNP there are 3 genotypes: – a diploid homozygote (two wild type alleles (PP)), a diploid homozygote (two mutant alleles (pp)) and a heterozygote (one wild type allele and one mutant allele (Pp)).
SNPs (single nucleotide polymorphisms) help predict an individual’s resistance or susceptibility to certain drugs (pharmacogenetics), susceptibility to environmental factors such as toxins, and risk of developing diseases. SNPs can also be used to track the inheritance of disease-associated genetic variants within families.
qPCR (real-time PCR) enables you to screen known SNPs. The benefits of qPCR are that it is easy, accurate, and can scale to high throughput. The bioinformatic analysis is also less complex than for other technologies, such as sequencing and microarrays.
5′-nuclease (TaqMan) allelic discrimination uses a primer pair to amplify the target area and two allele-specific probes to detect your target SNP alleles and report the genotypes of your samples. Unlike normal qPCR, in genotyping the fluorescence level is only measured at the end of the PCR and the results are displayed on a scatter plot, allowing hundreds of assays to be run and analysed in parallel.
Fecal matter is a highly complex, heterogenous material that contains PCR inhibitors including bile salts, polysaccharides, hematin and catabolic substances.
Yes, the Lyo-Ready™ Genotyping Direct qPCR Stool can be used as a liquid mix or lyophilized and stored at ambient temperature without needing to change the reaction conditions and lyophilization/storage will not affect the sensitivity of the test.
SNP biomarkers have been used for the detection of gastric, pancreatic, and colorectal cancers.
There are several buffers on the market that can be used for stool testing, they include phosphate-buffered saline (PBS), Cary-Blair Medium, Stool Transport and Recovery (STAR) buffer and DNA/RNA Shield.
Get In Touch With A Specialist
Have questions about a product? Want to learn more about Meridian’s molecular or immunoassay reagent portfolio? We want to hear from you!