Choose a country to view content specific to your location
Home » Life Science » Products » Molecular Reagents » NGS Library Quantification
The NGS Library Quantification Kit is an optimized, robust qPCR kit containing pre-diluted standards to minimize pipetting errors and a pre-qualified P5 and P7 Illumina adaptor sequence primer mix to ensure reproducible and precise qPCR results and an optimized buffer for dilution of NGS library samples.
Contact us to learn more about Meridian’s molecular or immunoassay reagent portfolio. We want to hear from you!
qPCR-based assay for quantification of only adapter-ligated NGS library molecules, thereby enabling optimal flow cell loading for maximum data yield and quality.
Reliable quantification of even low-yield libraries, ideal for both PCR and PCR-free library preparation methods.
Delivers fast, accurate assay results in as little as 90 minutes, thereby reducing time to results.
Contains a series of six pre-diluted DNA standards for rapid, simple standard curve development.
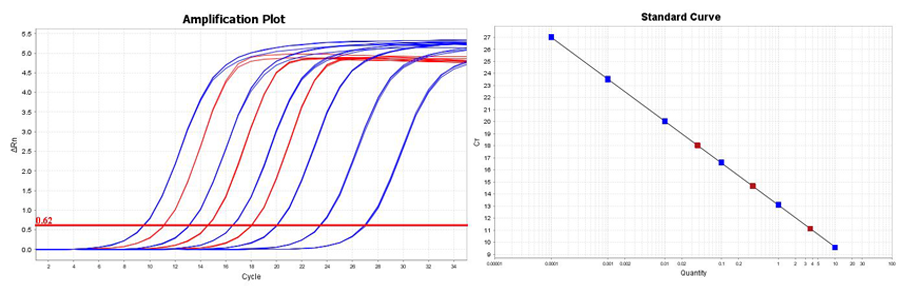
qPCR amplification of each of the six supplied DNA standards (blue) and a tenfold serial dilution of the diluted Illumina NGS library (red) were carried out with the supplied primer sets in triplicate. From the resulting standard curve, a size adjusted library concentration was then determined. The amplification plots demonstrate a limit of detection of 0.0001 pM (100 aM).
Fast qPCR Mix, pre-diluted DNA standards, primer mix and dilution buffer, designed to accurately measure the number of amplifiable DNA fragments in Illumina-based NGS libraries before loading into a flow cell.
| Description | Fast and robust quantification of Illumina-based NGS libraries. It consists of a SYBR®-based Fast qPCR Mix, pre-diluted DNA standards, primer mix and dilution buffer, designed to accurately measure the number of amplifiable DNA fragments before loading into a flow cell. |
| Concentration | 2x |
| Format | Clear, colorless solution |
| Hot Start | Antibody mediated |
| Application | qPCR, two-step RT-qPCR |
| Sample type | cDNA, DNA |
| Presentation | 9 vials |
| Storage | -20 °C |
| Mix stability | See outer label |
| Consistency | ± 0.5 Ct variance between test and reference sample |
| DNA Contamination | None detected in PCR amplification with traces overlay with the negative control on E. coli and mouse genomic DNA specific targets. |
| DNase Contamination | No detectable degradation |
Glycerol-Free T4 DNA Ligase (HC)
High-Specificity Pfu HS Catalog
Next-Generation Sequencing (NGS)
With its greater sensitivity and broad dynamic range, qPCR is seen as the gold standard for NGS library quantification, as it accurately measures the number of molecules that can serve as templates during library and cluster amplification, even with very dilute libraries.
The NGS Library Quantification Kit is an optimized, robust qPCR kit containing pre-diluted standards to minimize pipetting errors and a pre-qualified P5 and P7 Illumina adaptor sequence primer mix to ensure reproducible and precise qPCR results and an optimized buffer for dilution of NGS library samples.
No. However, the Library Quantification Kit contains primers which are compatible with P5 and P7 adaptor sequences specific to the Illumina platform.
Yes, the prepared library has to be diluted prior to running qPCR. Generally, we recommend preparing a 1:10,000 1:100,000 and 1:1,000,000 diluted sample of the library using the Dilution Buffer provided. Using two or more different dilutions of the library will ensure that at least one dilution falls within the dynamic range of the standard curve generated. This could be especially useful when the library concentration is high.
Instruments like the Qubit, spectrophotometer and Bioanalyser measure the total amount of DNA present in solution. The Library Quantification Kit will only quantify molecules with both adapters present. If the concentration measured by qPCR is significantly lower than what is measured by another methods, this suggests that only a small proportion of molecules correctly incorporated the adapters during adapter ligation.
Yes, we always recommend running triplicate qPCRs, as qPCR is an extremely sensitive measurement technique that is vulnerable to variation arising from liquid handling, instrument performing and sampling error.
Yes. The intercalating dye used in the kit binds DNA in proportion to the number of base pairs per double-stranded DNA product, so it is necessary to correct for product length by normalizing to the size of the DNA standard, 342 bp. The formular for doing this correction can be found in the Product Handling Guide.
Have questions about a product? Want to learn more about Meridian’s molecular or immunoassay reagent portfolio? We want to hear from you!
By submitting your information in this form, you agree that your personal information may be stored and processed in any country where we have facilities or service providers, and by using our “Contact Us” page you agree to the transfer of information to countries outside of your country of residence, including to the United States, which may provide for different data protection rules than in your country. The information you submit will be governed by our Privacy Statement.
| Specimen Type |
|---|
| Application |
| Catalog |
| Sizes |
| Wet(Liquid) |
| Air-Dryable |
| Glycerol-Free |
| Lyo-Ready |
| Concentration |
| High Concentration |
| Description |
| Request Sample |