Air-Dryable™ Direct DNA and RNA/DNA qPCR Urine Master Mixes
Air-Dryable™ Direct DNA qPCR and RNA/DNA qPCR Urine mixes are glycerol-free mixes that contain optimized excipients compatible with air and oven drying. The mixes have been designed for the development of quantitative PCR urine test assays for direct detection of DNA and RNA from human urine specimens without extraction. Can also be used wet and/or with purified nucleic acid
Have questions about a product?
Meridian's Air-Dryable™ Direct DNA and RNA/DNA qPCR Urine Master Mixes
- Create your ultra-sensitive multiplex direct quantitative PCR urine test using crude urine samples.
- Simplify qPCR urine testing, by removing the nucleic acid extraction step or inhibitor carryover during purification, shortening sample-to-result and reproducibility.
- Designed for the direct detection of bacteria and cell-free nucleic acids at very low titers.
- Can be used wet or it can be dried, for developing an ambient-temperate stable assay.
High performance (sensitivity and reproducibility) in the presence of 5% human urine
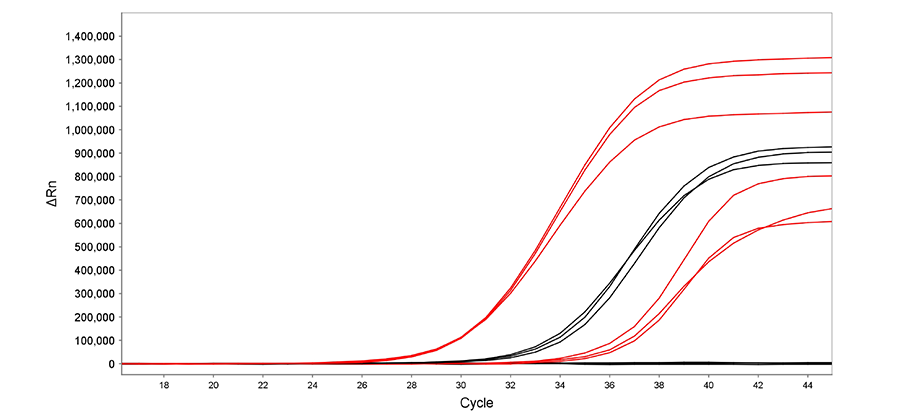
Activity of air-dried Air-Dryable™ Direct RNA/DNA qPCR Urine (red) was compared to Reliance 1-Step Multiplex RT-qPCR Supermix (purple) for their ability to amplify a 1,000-fold dilution of an RNA target (10,000 and 10 copies respectively) in the presence of 5% human urine. The results illustrate that the dried mixes maintain the same high performance as the liquid mixes and shows much higher sensitivity than Reliance 1-Step Multiplex RT-qPCR Supermix.
Air-Dryable Direct DNA qPCR Urine, 4x, MDX150
Inhibitor tolerance to 60% urine. Perfect for multiplexing and fast cycling conditions. Used in the detection of urinary track infections, STD, tropical diseases, cancer screening, bladder and prostate cancer.
Documents & Resources
Air-Dryable Direct RNA/DNA qPCR Urine, 4x, MDX151
Inhibitor tolerance to 20% urine. Perfect for multiplexing and fast cycling conditions. Used in the detection of urinary track infections, STD, tropical diseases, cancer screening, bladder and prostate cancer.
Documents & Resources
Description
Urine is an ideal clinical specimen because it is excreted in large quantities is non-invasive, and it can be self-sampled. Currently, urine specimens are used in the diagnosis and management of infectious diseases (including STDs), hormone and metabolic disorders, renal diseases, bladder cancer, urinary tract infections (UTIs), and for monitoring recreational drug use. However, urine contains substances such as urea and nucleases that can damage DNA or inhibit the PCR reaction.
Meridian’s new Air-Dryable™ Direct Urine molecular mixes are unique in that they enable the highly sensitive, very fast detection of target DNA or RNA directly from high concentrations of urine and are sensitive enough to detect arboviruses, such as Chikungunya virus. These mixes have been formulated specifically to overcome the inhibitors found in urine – no further optimization is required aside from the addition of primers and probes. Furthermore, these mixes can be used in a wet format or dried down by oven or air drying to create ambient-temperature stable assays.
Specifications
| Description | Glycerol-free qPCR and RT-qPCR mix containing Taq polymerase, reverse transcriptase, reaction buffer, dNTPs, MgCl2 and air-dry compatible excipients, developed to tolerate the effects of the inhibitors present in urine samples. |
| Concentration | 4x |
| Appearance | Clear, colorless solution |
| Hot Start | Antibody mediated |
| Application | Probe-based, real-time PCR, two-step RT-qPCR, one-step RT-qPCR |
| Sample type | cDNA, crude or purified RNA and/or DNA from human stool |
| Presentation | 1 vial |
| Storage | -20 °C |
| Mix stability | See outer label |
| Assay stability | Up to 24 months at ambient temperature following air-drying. |
| Consistency | ± 0.5 Ct variance between test and reference sample |
| DNase/RNase Contamination | No detectable degradation |
Catalogs & Brochures
Related Products
FAQs: qPCR Urine Test
What are the advantages of using urine over other sample types?
The advantages of a quantitative PCR urine test are that it is completely non-invasive, and it can be self-sampled in large quantities. Urine testing is used in the diagnosis and management of infectious diseases (including STDs), hormone and metabolic disorders, renal diseases, bladder cancer, urinary tract infections (UTIs) and for monitoring recreational drug use.
What types of infectious diseases can urine detect?
The most common infections are urinary tract infections (UTIs), most commonly Escherichia coli, Protus mirabilis, Enterococcus faecalis, Staphylococcus saprophyticus and Klebsiella pneumoniae. Several other infections can be diagnosed by urine DNA and urine RNA tests including community-acquired pneumonia (CAP), legionellosis, tuberculosis, congenital cytomegalovirus (CMV) infection, and arboviruses, such as Chikungunya virus, dengue virus and Zika virus. Parasites can also be diagnosed from urine testing for the detection of eggs from Schistosoma haematobium. Furthermore, urine has been used for screening of different sexually transmitted diseases (STD) such as Neisseria gonorrhoeae and Chlamydia trachomatis.
What type of cancer can be detected in urine?
Cancer cells or their DNA or RNA are too big to pass through the kidneys and will have to come from the bladder or ureters, so bladder cancer is the most obvious urine cancer, however prostate and cervical cancer are also detectable.
What is the maximum volume of urine you can use in a quantitative PCR urine test?
We have used up to 25% human urine with our mix.
After drying for how long will the mix stay stable to use? Or do we need to use it immediately after drying?
It is stable for up to 2 years at ambient temperature if correctly stored in sealed pouches.
Is a pre-treatment needed if you want to detect bacteria in urine?
That will depend on the bacteria, for some bacteria you may not need to do any pre-treatment, however some may require heating to 95°C for 5 min and for some tougher bacteria, a Proteinase K step (Proteinase K 55°C for 15 min and then at 98°C for 5 min) or using a non-ionic detergent (such as Tween-20 to final concentration of 0.2% followed by incubation at room temperature for 10 min) may be necessary.
What is the performance difference for DNA when using the DNA-specific urine mix versus the DNA/RNA urine mix?
We do not see a performance difference between the two mixes, which is why we call the Air-Dryable Direct RT-qPCR Urine mix – Air-Dryable™ Direct RNA/DNA qPCR Urine mix, as it can be used for both.
Are reaction conditions the same for air-dried and liquid mixes?
Yes, the Air-Dryable™ Direct DNA qPCR Urine and Air-Dryable™ Direct RNA/DNA qPCR Urine can be used as a liquid mix or air/oven dried and stored for up to 24 months without needing to change the reaction conditions and air drying/ambient temperature storage will not affect the sensitivity of the test.
Get In Touch With A Specialist
Have questions about a product? Want to learn more about Meridian’s molecular or immunoassay reagent portfolio? We want to hear from you!
By submitting your information in this form, you agree that your personal information may be stored and processed in any country where we have facilities or service providers, and by using our “Contact Us” page you agree to the transfer of information to countries outside of your country of residence, including to the United States, which may provide for different data protection rules than in your country. The information you submit will be governed by our Privacy Statement.