IDkit Hp® One
Effective immediately, the BreathID Hp instrument is discontinued. The purchase of kits and consumables will continue to be available including technical support for current instruments in use.
Catalog Number: AC00051
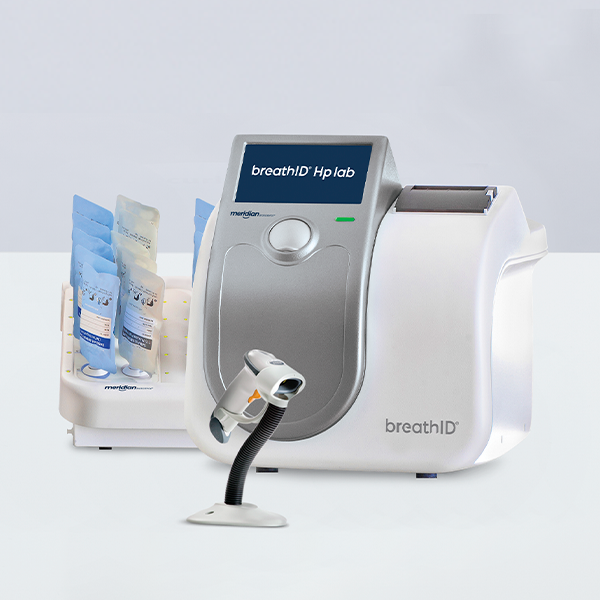

Continous flow specimen collection allows for improved patient satisfaction and compliance.
For use with BreathID® Hp
Related Tests
Gastrointestinal

Kit Components
-
Nasal cannula
-
75mg 13C-urea tablet
-
4.3g package of Citrica (citric acid)
-
Drinking straw
-
Package insert
Support & Documents
Downloadable PDFs
FAQs
What is the suggested CPT Code
H. pylori urea breath test analysis — 83013
H. pylori urea breath test collection — 83014
Pricing
To order, contact your regional sales representative or use the following contact information:
- Email: sales@meridianbioscience.com
- Phone: +1 800 696-0739
- International Sales Extension: +1 513 271-3000
